 
This indicates that the empirical formula of this compound i s not the same as its molecular formula.ĭivide 176.1 by 88 to see how many times larger the molar mass of the molecular formula is compared to the one of the empirical formula: For this, calculate the molar mass of the empirical formula C 3H 4O 3, to see if it matches the actual molar mass given in the problem (176.1 g/mol):ģ x 12.0 + 4 x 1.00 + 3 x 16.0 = 88 g/mol 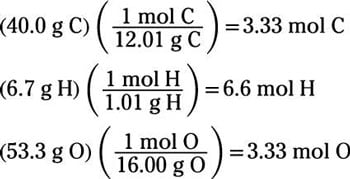
Therefore, by multiplying all the subscripts by 3, we get that the empirical formula isĤ) Next, we need to check if the empirical formula is the same as the molecular formula of the compound. Make sure to multiply all the subscripts – you cannot multiply only one and leave the others because that would change the mole ratio of the elements. Multiplying 1.343 by 3 is 4.029, which is approximately equal to 4 (4.029 ≈ 4). Remember, you can divide or multiply by any number to get a set of the simplest integers. We still need to simplify the moles of H. \ĭon’t worry about significant figures here and keep at least three decimals.ģ) Dividing the numbers by the smallest one is usually a good start to simplifying them. The molar mass of sulfuric acid is: M (H 2SO 4) = 2 x 1.0 + 32 + 4 x 16.0 = 98 g/mol. If the element is not specified, we need to calculate the percentage of all the elements. To calculate the percentage of an element, multiply its molar mass and the subscript which shows how many moles of it are present in one mole of the compound, then divide the product by the molar mass of the compound.įor example, let’s determine the percent composition of sulfuric acid (H 2SO 4). 
Percentage composition shows the amount of each element in a compound expressed as a mass percent. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
